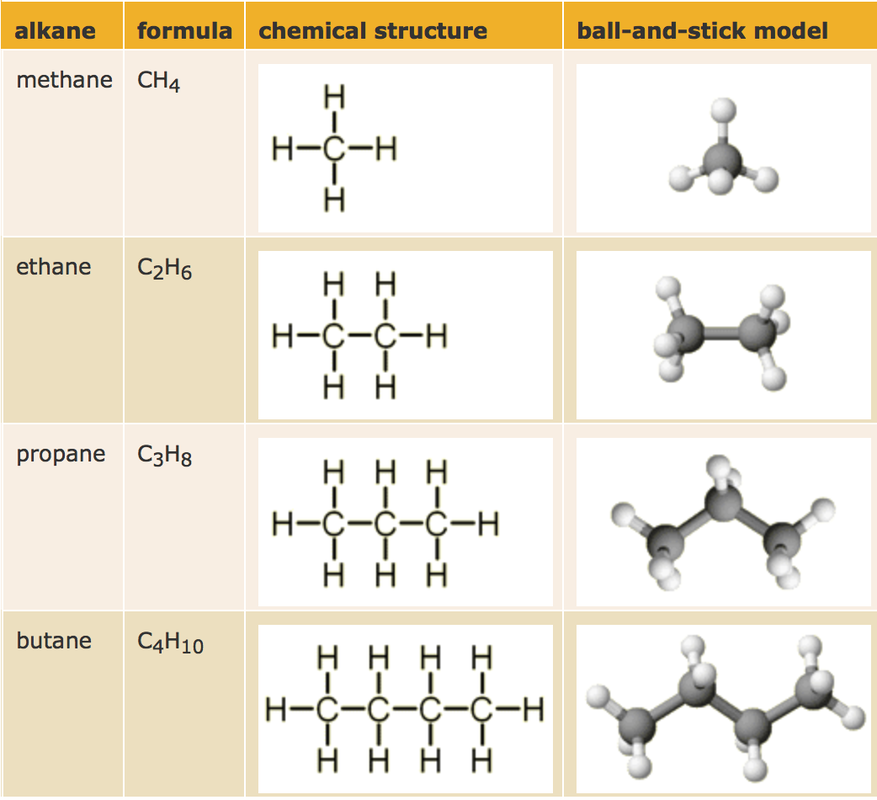
dISPLAYED FORMULA AND MOLECULAR
|
The number of hydrogen atoms in an alkane is double the number of carbon atoms, plus two.
For example, the molecular formula of methane is CH4. For ethane, it is C2H6. Alkane molecules can be represented by displayed formulae in which each atom is shown as its symbol (C or H), and the covalent bonds between them by a straight line. Alkanes are saturated hydrocarbons. This means their carbon atoms are joined to each other by single covalent bonds. |
|