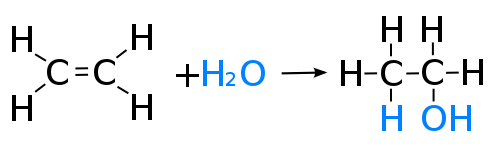1) hydration of ethene to form ethanol
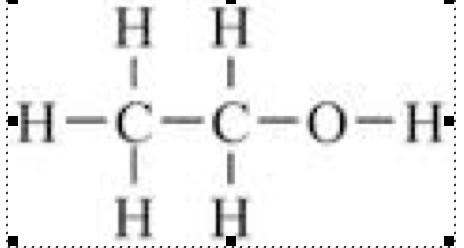
- Ethene + water --> ethanol
- C2H4+H20--> C2H5OH
- This is an addition reaction
- C2H4+H20--> C2H5OH
- This is an addition reaction
Factors that you need to consider
-300 °C, 60-70 atmospheres of pressure and phosphoric acid as catalyst. This means a lot more energy needed.
-Countries where land is scarce or not easy to cultivate and have oil reserves, hydration of ethane will be the favoured process
- Rate of reaction is fast
- Rate of production: Continuous process, keeps going
- Ethanol is the only product (pure); a higher yield.
-300 °C, 60-70 atmospheres of pressure and phosphoric acid as catalyst. This means a lot more energy needed.
-Countries where land is scarce or not easy to cultivate and have oil reserves, hydration of ethane will be the favoured process
- Rate of reaction is fast
- Rate of production: Continuous process, keeps going
- Ethanol is the only product (pure); a higher yield.
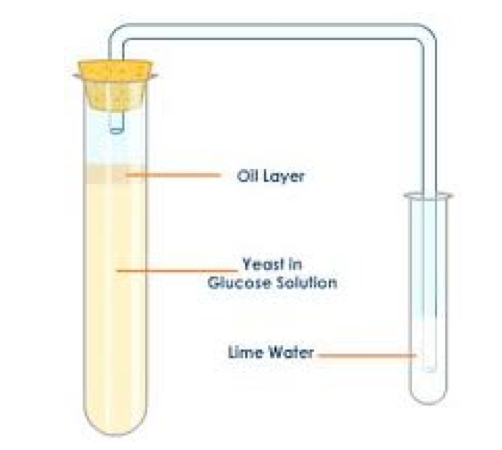
2)Fermintation
|
Fermentation is the reaction when sugar is changed into ethanol and carbon dioxide by yeast (single celled organisms). Yeast has the necessary enzyme which could act as a (biological catalyst) = speed up chemical reactions
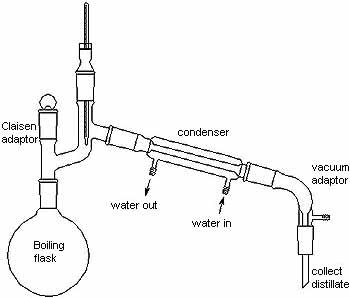
This reaction is also known as anaerobic respiration as no oxygen is involved in the reaction. Respiration is a reaction that gives out energy so the yeast ferments the sugar to obtain energy for its life processes; carbon dioxide and ethanol are the waste products of this reaction. The ethanol can be obtained through fractional distillation of the mixture. The equations for fermentation are: balanced symbol equation C6H12O6 (aq) ---> 2C2H5OH (aq) + 2CO2 (g) word equation: enzyme in yeast sugar (e.g. glucose) ----> ethanol + carbon dioxide Conditions: sugar in water, yeast and a temperature of 30 °C to 40 °C. As yeast is a living organism the optimum temperature for fermentation is 35 °C. At higher temperatures the reaction nearly stops as the enzyme is broken down or denatured. |