Addition reaction
An addition reaction is a reaction during which a small molecule is added to an unsaturated organic compound changing it into a saturated compound.
This happens because the double bond pops open and each carbon electron of the double bond can now be used to make a new covalent bond with another atom or group of atoms.
The small molecule which reacts with the alkene breaks up into 2 parts and one part goes to one carbon of the double bond and the other part goes to the second carbon atom of the double bond.
examples:
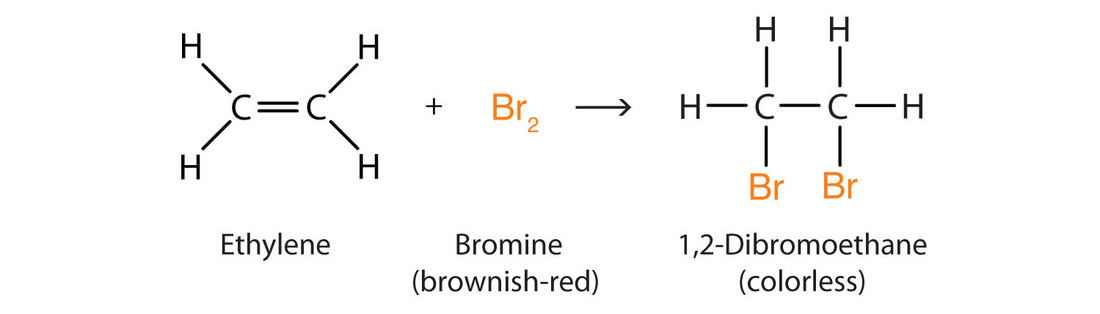
alkene reacting with bromine-
ethene + bromine
An addition reaction is a reaction during which a small molecule is added to an unsaturated organic compound changing it into a saturated compound.
This happens because the double bond pops open and each carbon electron of the double bond can now be used to make a new covalent bond with another atom or group of atoms.
The small molecule which reacts with the alkene breaks up into 2 parts and one part goes to one carbon of the double bond and the other part goes to the second carbon atom of the double bond.
examples:
alkene reacting with bromine-
ethene + bromine
- The double bonds break in the ethylene this means that it is now a single bond, the Br2 is then added onto the bond that was broken in the Ethylene
- molecular formula: C3H6+Br --> C3H6Br2