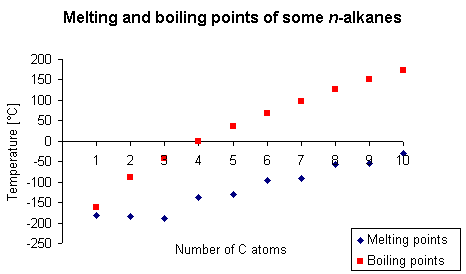
Trends in physical properties
The alkanes can exist as gases, liquids, or solids at room temperature. The unbranched alkanes methane, ethane, propane, and butane are gases; pentane through hexadecane are liquids; the larger the homologues than the hexadecane are solids.
As the numbers of carbon increase, molecular mass also increases. This increases the intermolecular forces which require more energy to be broken. As a result melting and boiling points also increase
As the numbers of carbon increase, molecular mass also increases. This increases the intermolecular forces which require more energy to be broken. As a result melting and boiling points also increase
- The branched alkanes usually have lower boiling points
- Solid alkanes are normally soft and has low melting points, this is because of the strong repulsive forces that are generated between the electrons on the neighboring atom
- Alkanes are mostly insoluble in water